
Evidence review: N-acetylcysteine for OCD [Updated March 2026]
Introduction
N-acetylcysteine (NAC) is thought to regulate glutamate neurotransmission in the brain, and also acts as a potential anti-oxidant. It increases the amount of extracellular glutamate by enhancing the activity of the cystine/glutamate transporter.
There are a number of other drugs that are sometimes used to treat OCD that act on glutamate. These include: Memantine; Riluzole, D-cycloserine; and Lamotrigine. The possible mechanism of action is unknown, but there is evidence that glutamate is involved in development of the symptoms of obsessive-compulsive disorder (OCD).[1-3] Of course, glutamate is the main 'excitatory' neurotransmitter in the brain, so it is likely to be involved in all neural processes to some extent.
Parts of the brain that are likely to be involved in the development of OCD (such as the striatum, parts of the basal ganglia, and some of the cortico-striatral-thalamo-cortical circuits) have large amounts of excitatory (i.e. glutamatergic) input relating to information about movements and sensory integration. It is possible that this large amount of glutamatergic input puts these parts of the brain at risk of becoming dysregulated, with overstimulation of extra-synaptic glutamate receptors.
Reason for update
There have been several recent systematic reviews of NAC published since the previous summary.[4, 5] One (Eghdami et al, 2024) reported that: “The results of our study indicated a positive outcome for the experimental group in terms of the total Y-BOCS score when using the medication for a period of five to eight weeks (p-Value = 0.05). However, no significant difference was observed for durations shorter than five weeks or longer than 12 weeks.” The other (Gadallah et al, 2020) concluded: “Regarding the Yale-Brown Obsessive Compulsive Scale, which was the primary outcome, the overall effect size favored NAC (MD = −2.97, 95% CI [-4.93, −1.02], P = 0.003).” There has also been correspondence published addressing both some of the positive results but also the uncertainty regarding NAC as a treatment for OCD.[6]
Methods
This is not a systematic review, but is based around a review of what information is available. It is intended to provide an overview of what the evidence would support in terms of treatment. Only randomised, controlled studies (RCTs) have been included.
Outcome data (intervention vs control) were extracted and entered into ‘forest plots’ showing the outcomes from each study, along with the combined effect of all studies combined. These forest plots were drawn using the MS Excel files from Exploratory Software for Confidence Intervals, a resource developed alongside a book by Geoff Cumming. Intention-to-treat outcomes were extracted where available.
In one study,[4] data had to be extracted using WebPlotDigitizer.[5]
Results
Seven RCTs of N-Acetylcysteine have been included.[7, 9-13] A summary of these trials is given below in Table 1. The studies are sorted by year (earliest first).
Five studies were in adults, whilst two studies were in patients under the age of 18 years.
The average baseline Y-BOCS score across studies is 24.6. This means that patients had clinically significant symptoms and would be classed as ‘severe’ on the Y-BOCS, but patients being seen in specialist mental health services will probably have slightly higher scores and will have had more extensive treatment.
Table 1:
| Study ID | Place conducted | No. of participants |
Population |
Duration (weeks) |
Intervention vs Control |
Baseline Y-BOCS (mean ± SD) |
Age ± SD |
Other current treatment |
Previous failed treatment |
Primary outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Afshar, 2012 |
Iran |
48 |
Adult |
12 |
N-acetylcysteine (up to 2400mg/day) vs placebo |
27.7 ± 5.5 |
30.9 ± 5.0 |
SRI |
12 weeks of SSRI or Clomipramine |
Y-BOCS |
| Sarris, 2015 |
Australia |
44 |
Adult |
16 |
N-Acetylecystine (3000mg/day) vs placebo |
26.6 ± 5.7 |
37 (range 18-68) |
70% were taking medication; 57% taking SSRIs |
No treatment, or stable medication |
Y-BOCS |
| Paydary, 2016 |
Iran |
44 |
Adult |
10 |
N-Acetylcystine (2000mg/day) vs placebo |
27.0 ± 4.4 |
33.7 ± 11.3 |
Fluvoxamine 200mg/day |
None for 6 weeks prior to study |
Y-BOCS |
| Costa, 2017 |
Brazil |
40 |
Adult |
16 |
N-Acetylcysteine (3000mg/day) vs placebo |
25.2 ± 3.9 |
38.0 ± 10.8 |
Stable dose of SRI |
Failure to respond to at least 1 adeuqate drug treatment (Clomipramine or SSRI) |
Y-BOCS |
| Ghanizadeh, 2017 |
Iran |
34 |
Child & Adolescent |
10 |
N-Acetylcysteine (2400mg/day) vs placebo |
21.0 ± 8.2 |
16.5 ± 2.9 |
SRI |
At least a previous trial of an SSRI |
Y-BOCS |
| Li, 2020 |
USA |
11 |
Child & Adolescent |
12 |
N-Acetylcysteine (2700mg/day) vs placebo |
21.4 ± 4.7 |
13.4 ± 3.4 |
Unspecified amount on SRI |
None specified |
CY-BOCS |
| Sarris, 2022 |
Australia |
98 |
Adults |
20 |
N-Acetylcysteine (2000-4000mg/day) vs placebo |
23.1 ± 4.0 |
31.5 ± 20.8 |
SSRI |
1 failed trial (45%), 2 failed trials (25%), More than 3 failed trials (30%) |
Y-BOCS |
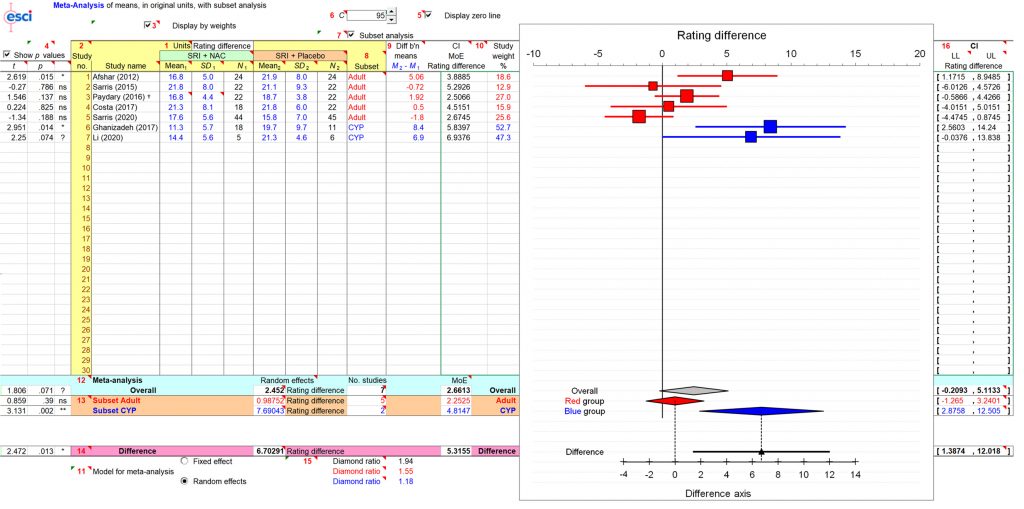
Forest plot of outcomes
Forest plots, showing the estimated results from each study along with the overall results, are shown below. Groups have been defined by whether they were in adults or children/ adolescents.
Overall, when all studies are combined there was no significant effect for NAC vs placebo, and in adults the lack of effect is much clearer. In the two trials of NAC in children and adolescents there is a positive effect.
It should be recognised that 4/5 of the adult studies were not statistically significant. Removing the single positive study (Afshar et al, 2012) reduces the (already negative) effect for NAC even further.
Updated findings
Compared to the previous summary, one additional RCT has been included (Sarris et al, 2022). This trial did not find a significant difference between NAC and placebo, and it was the largest trial included, so it means that there is no clear difference between NAC and placebo for adults.
Conclusions
If N-Acetylcysteine (NAC) has an effect, it is likely to be small and only evident in adolescents. There is no convincing effect for NAC in adults.
The observation that the first trial is positive, whilst successive trials are negative, is commonly seen in science.
The majority of studies included individuals who were not particularly severe. The non-weighted average baseline Y-BOCS across all studies was 24.8, and in adult populations only it was 26.6. This is broadly similar to most other trials of psychological/ drug treatment for OCD.
In general, tolerability of N-Acetylcysteine was good. In most studies, there were no significant differences between the NAC group and the placebo group. The most common adverse effects include: nausea; changes in bowel habit; and headache. Most side effects tend to be mild, and few studies report drop-outs. It often has an unpleasant smell that can make it difficult to take.
N-Acetylcysteine can be found in health-food shops and is typically around £7 for 30 capsules at a dose of 600mg. This means that to get up to the typical doses used in the studies, 30 capsules will last you about one week. Larger quantities can be bough online but the dose of capsules is the same, so 180 capsules will last you just over a month.
Overall, it is likely to be safe for most adults, but it is unlikely to have a clinically significant benefit; particularly in those with more severe symptoms and those who have not responded to at least one or two previous treatments. However, some people may wish to try it given its ‘health food’ status and its good tolerability.
References
- KARIUKI-NYUTHE C, GOMEZ-MANCILLA B, STEIN DJ. Obsessive compulsive disorder and the glutamatergic system. Current Opinion in Psychiatry. 2014; 27(1): 32–37. http://dx.doi.org/10.1097/YCO.0000000000000017
- PITTENGER C, BLOCH MH, WILLIAMS K. Glutamate abnormalities in obsessive compulsive disorder: neurobiology, pathophysiology, and treatment. Pharmacology & Therapeutics. 2011; 132(3): 314–332. https://doi.org/10.1016/j.pharmthera.2011.09.006
- VLCEK P, POLAK J, BRUNOVSKY M, HORACEK J. Role of Glutamatergic System in Obsessive-Compulsive Disorder with Possible Therapeutic Implications. Pharmacopsychiatry. 2018; 51(6): 229–242. https://doi.org/10.1055/s-0043-118665
- EGHDAMI S, EISSAZADE N, HEIDARI MOKARAR M, BOROON M, ORSOLINI L, SHALBAFAN M. The safety and efficacy of N-acetylcysteine as an augmentation in the treatment of obsessive-compulsive disorder in adults: a systematic review and meta-analysis of randomized clinical trials. Frontiers in Psychiatry. 2024; 15: 1421150. https://doi.org/10.3389/fpsyt.2024.1421150
- GADALLAH A-HA, EBADA MA, GADALLAH A, AHMED H, RASHAD W, EID KA, BAHBAH E, ALKANJ S. Efficacy and safety of N-acetylcysteine as add-on therapy in the treatment of obsessive-compulsive disorder: A systematic review and meta-analysis. Journal of Obsessive-Compulsive and Related Disorders. 2020; 25: 100529. https://doi.org/10.1016/j.jocrd.2020.100529
- CAROLLO M, CAROLLO N, MONTAN G. The promise of N-acetylcysteine in the treatment of obsessive-compulsive disorder. CNS Neuroscience & Therapeutics. 2024; 30(2): e14653. https://doi.org/10.1111/cns.14653
- PAYDARY K, AKAMALOO A, AHMADIPOUR A, PISHGAR F, EMAMZADEHFARD S, AKHONDZADEH S. N-acetylcysteine augmentation therapy for moderate-to-severe obsessive-compulsive disorder: randomized, double-blind, placebo-controlled trial. Journal of Clinical Pharmacy and Therapeutics. 2016; 41(2): 214–219. https://doi.org/10.1111/jcpt.12370
- WebPlotDigitizer [Computer Program] (2020). Version 4.3 for Microsoft Windows. Pacifica, California, USA: Automeris LLC. https://automeris.io/WebPlotDigitizer.
- AFSHAR H, ROOHAFZA H, MOHAMMAD-BEIGI H, HAGHIGHI M, JAHANGARD L, SHOKOUH P, SADEGHI M, HAFEZIAN H. N-Acetylcysteine Add-On Treatment in Refractory Obsessive-Compulsive Disorder: A Randomized, Double-Blind, Placebo-Controlled Trial. Journal of Clinical Psychopharmacology. 2012; 32(6): 797–803. https://doi.org/10.1097/JCP.0b013e318272677d
- COSTA DLC, DINIZ JB, REQUENA G, JOAQUIM MA, PITTENGER C, BLOCH MH, MIGUEL EC, SHAVITT RG. Randomized, Double-Blind, Placebo-Controlled Trial of N-Acetylcysteine Augmentation for Treatment-Resistant Obsessive-Compulsive Disorder. Journal of Clinical Psychiatry. 2017; 78(7): e766–e773. https://doi.org/10.4088/JCP.16m11101
- GHANIZADEH A, MOHAMMADI MR, BAHRAINI S, KESHAVARZI Z, FIROOZABADI A, ALAVI SHOSHTARI A. Efficacy of N-Acetylcysteine Augmentation on Obsessive Compulsive Disorder: A Multicenter Randomized Double Blind Placebo Controlled Clinical Trial. Iranian Journal of Psychiatry. 2017; 12(2): 134–141. https://www.ncbi.nlm.nih.gov/pubmed/28659986
- LI F, WELLING MC, JOHNSON JA, COUGHLIN C, MULQUEEN J, JAKUBOVSKI E, COURY S, LANDEROS-WEISENBERGER A, BLOCH MH. N-Acetylcysteine for Pediatric Obsessive-Compulsive Disorder: A Small Pilot Study. Journal of Child and Adolescent Psychopharmacology. 2020; 30(1): 32–37. https://doi.org/10.1089/cap.2019.0041
- SARRIS J, OLIVER G, CAMFIELD DA, DEAN OM, DOWLING N, SMITH DJ, MURPHY J, MENON R, BERK M, BLAIR-WEST S, NG CH. N-Acetyl Cysteine (NAC) in the Treatment of Obsessive-Compulsive Disorder: A 16-Week, Double-Blind, Randomised, Placebo-Controlled Study. CNS Drugs. 2015; 29(9): 801–809. https://doi.org/10.1007/s40263-015-0272-9
Last Updated on 31 March 2026 by David Christmas

Leave a Comment
(1 Comment)
[…] You're reading the older version of this review. Please visit the updated version. […]
Recent posts
Tags